Why You Don’t Use the DOT Emergency Response Guidebook for Incidents Involving Anhydrous Ammonia at a Fixed Facility!

My intent with this FFN discussion is to educate and warn readers that you need to always have at least two reference sources and preferably three or more when making a determination about a chemicals properties and how to mitigate an incident.
This discussion focuses on Anhydrous Ammonia because firefighters continue to get injured or killed in facilities using a gas that should be considered a flammable gas but in the United States, ammonia is considered a non-flammable gas. Take a look at most online MSDS sheets and they will list the flammable range for anhydrous ammonia as 16-25% which is data that was researched and published in the 1950's. Don't you think that our technology to test chemicals has gotten a little more sophisticated in the past 50 years? At firefighter's expense, the NFPA has turned their back on updating their data to reflect more recent testing data that provided more than sufficient data to document and prove that anhydrous ammonia is indeed a flammable gas. After readingt this post, you will know why.
For the most part, MSDS's that you look up on line will reflect the old standard which facilitates NH3 to be considered a non-flammable gas. However, I did find one MSDS that correctly addresses the flammable range as 15-28%, which by definition makes it a flammable gas. This company is honest and shares the most recent information for chemical flammability testing. Here’s the link:
http://www.wdserviceco.com/MSDSANHY.html
An important note to look at on the MSDS Sheet is the NFPA Hazard Diamond recommendation. The NFPA classifies anhydrous ammonia as a non-flammable gas, listing 15-25% for the flammable range. As a result, the NFPA hazard rating is listed as a 3-1-0. Why won’t the NFPA address the issue of anhydrous ammonia meeting the definition of a flammable gas? If you have access to the NIOSH Pocket Guide for Chemical Hazards, look up anhydrous ammonia and note that they show the flammable range as being 15-28%. This book is considered the bible for the latest and greatest permissible exposure levels, flammable ranges, etc. Industrial Hygenists get together annually and provide updated information for the various chemicals. Many of the lessons learned in regard to acceptable exposure levels were developed using firefighters as the guinnea pigs. When I first researched this over 15 years ago, I found out that one of the biggest reasons for not changing the classification of NH3 was the cost associated with upgrading facilities to a Class I Division 1 electrical installation requirement. The safety factors are not taken into consideration by the NFPA which is heavily supported by industry. It is very much the fox guarding the hen house...
Ammonia is a chemical consisting of one atom of nitrogen and three atoms of hydrogen. It is designated in chemical notation as NH3. Ammonia is extremely soluble in water and is frequently used as a water solution called aqua ammonia. Ammonia chemically combines with water to form ammonium hydroxide. Household ammonia is a diluted water solution containing 5 to 10 percent ammonia. On the other hand, anhydrous ammonia is essentially pure (over 99 percent) ammonia. "Anhydrous" is a Greek word meaning "without water;" therefore, anhydrous ammonia in ammonia without water.
Refrigerant grade anhydrous ammonia is a clear, colorless liquid or gas, free from visible impurities. It is at least 99.95 percent pure ammonia. Water cannot have a content above 33 parts per million (ppm) and oil cannot have a content above 2 ppm. Preserving the purity of the ammonia is essential to ensure proper function of ammonia refrigeration systems.
In 1984, one firefighter was killed and a second was burned over 72% of his body in an anhydrous ammonia explosion and fire that occurred in Shreveport, LA. Ammonia was leaking inside a cold storage building. While firefighters were working inside wearing Level A chemical protection and oxygen re-breather SCBA’s, the ammonia reached an ignition source. The fire department used the DOT ERG as a guide for how to mitigate the incident and it resulted in tragedy.
Though it is listed as a non-flammable gas by DOT, ammonia burns inside structures and confined spaces; it is less likely to ignite out in the open as long as the leaking vessel is outdoors. My hazmat team was responded to a leaking ammonia vessel in a town called Morro Bay. It was an annual festival weekend with lot's of money on the line. The town needed to be open for business. The problem was that not only was there potential for a fire or explosion but the leaking NH3 tank would produce some significant downwind exposure issues. For leaking tanks inside a structure, it is key to ensure that there are not ignition sources and that you ventilate to minimize the potential for an explosion. For those downwind from the incident, sheltering in place is usually the method of choice due to the logistics of moving people in a possible IDLH environment.
Precautions should be taken for ammonia leaks inside buildings just as for any other flammable gas. The Shreveport FD used the DOT Emergency Response Guidebook as a tool for mitigating an incident inside a structure. The results were both fatal and tragic. The DOT Emergency Guidebook was designed for highway incidents, not fixed facilities.
In the 2008 DOT Emergency Guidebook, Page 2, 2nd paragraph:
“This guidebook will assist responders in making initial decisions upon arriving at the scene of a dangerous goods incident. It should not be considered as a substitute for emergency response training, knowledge or sound judgment. ERG2008 does not address all possible circumstances that may be associated with dangerous goods incidents. It is primarily designed for the use at a dangerous goods incident occurring on a highway or railroad. Be mindful that there may be limited value in its application at fixed facility locations.”
When using the DOT Emergency Response Guidebook, if you were to use this as your reference source, this is what you will currently read.
DOT 2008 Emergency Response Guidebook
FIRE OR EXPLOSION
• Some may burn but none ignite readily.
• Vapors from liquefied gas are initially heavier than air and spread along ground.
• Some of these materials may react violently with water.
• Cylinders exposed to fire may vent and release toxic and/or corrosive gas through pressure relief devices.
• Containers may explode when heated.
• Ruptured cylinders may rocket.
What is a flammable gas?
Flammable Gas: A gas that, at an ambient temperature and pressure, forms a flammable mixture with air at a concentration of 13 percent by volume or less; or, a gas that, at an ambient temperature and pressure forms a range of flammable mixtures with air wider than 12 percent by volume, regardless of the lower limit.
Anhydrous ammonia is classified by the U.S. Department of Transportation (DOT) as a Class 2.2 Non-flammable Gas. Unfortunately, this classification leaves two important hazards of anhydrous ammonia unidentified by the DOT placarding and labeling system. Not only will anhydrous ammonia burn under certain conditions, it is classified as a caustic (corrosive) liquid and poison gas in other parts of the world. U.S. manufacturers identify the hazards as flammable, toxic and corrosive.
Anhydrous means without water. Other chemicals also have the word anhydrous in their name and it means the same thing, without water. Anhydrous ammonia (NH3) is a colorless liquefied gas that is free of water; for that reason, it has a high affinity for water. Thirteen hundred gallons of ammonia vapor will dissolve in just one gallon of water. It has a very sharp, intensely irritating odor anyone in the area of a release will not want to stay! Because of its great affinity for water, care must be taken in the storage and handling of ammonia to keep it dry. When ammonia gas is dissolved in water, the resulting material is ammonium hydroxide or “aqua” ammonia. The two materials should not be confused.
Ammonia gas is lighter than air and is easily liquefied by pressure. It has an auto ignition temperature of 1,204 degrees Fahrenheit and a flammable range of 16-25%. The reason the DOT does not consider ammonia a flammable gas is the definition used for flammable gases. According to the DOT, a flammable gas has a lower explosive limit (LEL) below 13 or a flammable range of greater than 12 percentage points.
Ammonia misses the definition on both counts using data from testing done years ago. According to current MSDS Data, Ammonia has a LEL of 16, three points above the DOT requirement for a flammable gas, and the flammable range is 10 percentage points, not the 12 required by the DOT’s definition. The reality is that anhydrous ammonia can and will burn, and has injured and killed firefighters when it ignited. Anhydrous ammonia is toxic with a threshold limit value (TLV) of 25 ppm in air. Inhalation of concentrated fumes may be fatal.
Has there been other incident involving Anhydrous Ammonia that hurt or killed firefighters?
There have been numerous explosions and fatal exposures at facilities that store, use and handle anhydrous ammonia. Ice Cream manufacturing plants, cold storage warehouses and blue print operations all use anhydrous ammonia in the processing.
An accident occurred in the late 1990’s in a cold storage building in Ortana, PA. Two plant maintenance workers, who were also local volunteer firefighters, were conducting routine maintenance on liquid ammonia lines within the facility. A leak occurred, the men were splashed with liquid ammonia and both died. Firefighting personnel responding to the incident were burned by ammonia vapors as they entered the facility in turnouts to rescue the workers. Not all incidents obviously result in an explosion but having a thorough understanding of how to take care of anhydrous ammonia victims is paramount toward increasing survival from this type of chemical exposure.

January 8, 2008 / Warehouse Blaze Kills 40
A devastating blaze Monday on the basement floor of a cold storage facility in Icheon, Gyeonggi Province, killed 40 of at least 57 working in the warehouse at the time. The remaining workers escaped or were rescued and have since been treated in hospital. Seven of them are in serious condition.
According to police and firefighting authorities, the fire started with an explosion in the machine room on the basement floor of the cold storage warehouse of the company, Korea 2000, in Icheon, Gyeonggi Province around 10:50 a.m. on Monday. When the fire broke out, it is believed that 57 workers were installing electric wiring or pumping Freon gas, a refrigerant for cold storage facilities, on the basement floor. The fire soon engulfed the entire building, giving off toxic gases.
Medical Considerations:
Chemical burns are associated with significant morbidity, especially anhydrous ammonia burns. Anhydrous ammonia is a colorless, pungent gas that is stored and transported under pressure in liquid form. A 28 year-old patient suffered 45% total body surface area of second and third degree burns as well as inhalational injury from an anhydrous ammonia explosion. Along with fluid resuscitation, the patient’s body was scrubbed every 6 hours with sterile water for the first 48 h to decrease the skin pH from 10 to 6–8. He subsequently underwent a total of seven wound debridements; initially with allograft and then autograft. On post burn day 45, he was discharged. The injuries associated with anhydrous ammonia burns are specific to the effects of ammonium hydroxide. Severity of symptoms and tissue damage produced is directly related to the concentration of hydroxyl ions. Liquefactive necrosis results in superficial to full-thickness tissue loss. The affinity of anhydrous ammonia and its byproducts for mucous membranes can result in hemoptysis, pharyngitis, pulmonary edema, and bronchiectasis. Ocular sequelae include iritis, glaucoma, cataracts, and retinal atrophy.
The desirability of treating anhydrous ammonia burns immediately cannot be overemphasized. Clothing must be removed quickly, and irrigation with water initiated at the scene and continued for the first 24 hours. Resuscitative measures should be started as well as early debridement of nonviable skin. Patients with significant facial or pharyngeal burns should be intubated, and the eyes irrigated until a conjunctivae sac pH below 8.5 is achieved. Although health care and emergency response professionals need to be prepared to treat chemical burns, educating the public, especially those workers in the agricultural and industrial setting, should be the first line of prevention.
Ammonia Releases Can Cause Major Evacuations:
The Tampa Fire Department dealt with a large ammonia incident and did an outstanding job communicating evacuation information using a local resource called TampaBay.com.
http://images.google.com/imgres?imgurl=http://blogs.tampabay.com/ph...">Click here for the TampaBay ammonia incident video.
Under normal conditions, ammonia is a very stable compound. It takes excessive temperatures (about 840° to 930°F) to cause it to dissociate slightly at atmospheric pressure. When this happens, the dissociated products are nitrogen and hydrogen. Ammonia gas burns in a mixture with air within a limited range. The flammable limits at atmospheric pressure are 15% to 28% by volume of ammonia in air. Experiments conducted by Underwriters Laboratories indicate that an ammonia-air mixture in a standard quartz bomb will not ignite at temperatures below 1562°F. When an iron bomb, having a catalytic effect, was used, the ignition temperature dropped to 1204°F.
Properties
Anhydrous ammonia is the compound formed by the combination of the two gaseous elements, nitrogen and hydrogen, in the proportion of one part of nitrogen to three parts of hydrogen by volume. Since one volume of nitrogen weighs fourteen times as much as one volume of hydrogen, on a weight basis, the ratio is fourteen parts of nitrogen to three parts of hydrogen, or about 82% nitrogen and 18% hydrogen.
At atmospheric temperature and pressures, anhydrous ammonia is a pungent colorless gas. Anhydrous ammonia boils at -28°F and freezes to a white crystalline mass at -108°F. When heated above its critical temperature of 270.3°F ammonia exists only as a vapor regardless of the pressure. Between the melting and critical points, liquid ammonia exerts a vapor pressure which increases with rising temperature. When liquid ammonia is in a closed container, it is in equilibrium with ammonia vapor and the pressure within the container bears a definite relationship to the temperature.
Liquid anhydrous ammonia is lighter than water, having a density of 42.57 pounds per cubic foot at -28°F, while as a vapor, ammonia is lighter than air, its relative density is 0.597 compared to air at atmospheric pressure and a temperature of 32°F. Under the latter conditions, one pound of ammonia vapor occupies a volume of 20.78 cubic feet. At 70°F and at atmospheric pressure, one pound of ammonia vapor occupies a volume of 22.5 cubic feet and yields 45 cubic feet of dissociated gas at a ratio of 25% nitrogen and 75% hydrogen.
Key Point: 75% Hydrogen… can you see why under the right conditions, inside a structure, and there is an ignition source, why Anhydrous Ammonia can and has behaved like the monster it is… a flammable gas.
Physical Constants of Anhydrous Ammonia
Molecular symbol: NH3
Molecular weight: 17.032
Boiling point at one atmosphere: -28°F
Freezing point at one atmosphere: -108°F
Critical temperature: 270.32°F
Critical pressure: 1657 psia
Vapor density at -28°F and one atmosphere: 0.056697 lb/cubic ft.
Heat of Combustion: 8001 BTU/lb
Flammable Limits: 15% to 28%
Ignition Temperature: 1204°F to 1562°F
Cheat Sheet for NFPA 704 Hazard Ranking
http://www.nmsu.edu/~safety/programs/chem_safety/NFPA-ratingA-C.htm
Planning for and Responding to Ammonia Incidents
http://www.mda.state.mn.us/chemicals/spills/ammoniaspills/fdrespons...
Tags:
Replies to This Discussion
-
Permalink Reply by Justin Buck on September 27, 2008 at 1:01am
-
you got a point. you bring up some go discussions on FFN.
-
Permalink Reply by Ben Waller on September 27, 2008 at 1:13am
-
Mike,
Even more importantly, anhydrous ammonia used as refrigerant is pumped around a plumbing system by a pump. That pump is lubricated with compressor oil, which is a liquid petroleum hydrocarbon, and in a fine mist/aerosol form. The fact that the ammonia is now fouled with a flammable aerosol makes the problem much worse. That is what happened in the Shreveport incident you cited. The firefighers were in an enclosed environment while wearing rubber suits.
It is fortunate that there were not more LODDs, given the circumstances.
-
Permalink Reply by Brian Dumser on September 27, 2008 at 1:19am
-
Excellent as usual Mike. I think a lot of us have come to rely on the ERG as the HAZMAT bible of sorts. We need to realize that it won't apply to every situation. Ben brings some good food for thought also with the Anhydrous Ammonia being fouled by the compressor oil. Things that make you go, "Hmmmmm."
-
Permalink Reply by Ben Waller on September 27, 2008 at 2:09am
-
Capt.,
I'd qualify your post with "...as long as the leaking vessel is outdoors."
A few years ago, my hazmat team responded to an anhydrous ammonia leak, from a vessel, but the vessel was inside a buttoned-up cold storage plant. The first responders sent three engine companies and a truck company (13 firefighters in all) to the hospital with varying degrees of chemical burns to the skin. They were inside doing a search for life in an atmosphere that would have been lethal to everyone in the structure prior to the first engine's arrival. None of them died, fortunately. We did the entry in Level A, shut down three valves, and ventilated. 20 minutes or so later, no ammonia cloud and no problem.
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:29am
-
Well, I wouldn't want to "divorce" myself from the ERG completely but when dealing with anything inside of a box, and specially when it's a compressed gas, you gotta figure that it's going to want to get out of the box and that even flour under the right circumstances can go boom. I spent a few days researching and composing this. I want to make sure folks learn from others fatal mistakes.
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:35am
-
Excellent points brother. Even with SCBA's in place, there are going to be a couple of molecules of ammonia to remind you what you are dealing with. Ammonia burns can be really nasty. These are very deep burns.

What's amazing is that by simply providing a minimum of 20-minutes of on scene flushing with tepid water, the ammonia is washed away from the skin and diluted to a point where it is no longer harmful. It's when this process is delayed that the injuries present themselves as being much more serious.
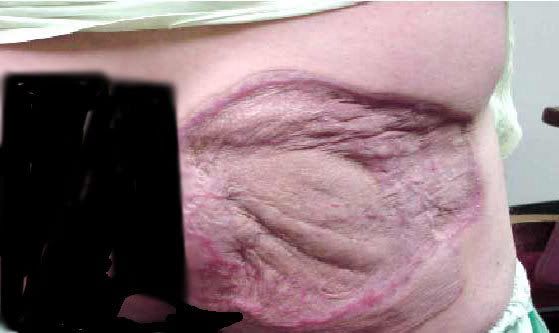
Anhydrous ammonia that makes contact with the skin causes both cold injuries as well as the toxic component that results in very deep burns.
-
Permalink Reply by T.J. on September 27, 2008 at 4:47am
-
Well put once again Mike, we've had a couple calls to our local farmers who have spilled anhydrous ammonia before not real large amounts but 20-40 gallons, nasty stuff it is, and I call I hate to hear come in.
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:50am
-
Additional language added to the post to clarify the leaking vessel indoors vs. outdoors scenario. What you did not share with your post was the fact that you ensured that there were not ignition sources. Before making entry into the building, does your department SOG's require you to identify to everyone over the radio on the incident that the power has been shut down? As always thanks for the input Chief. TCSS, Mike
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:51am
-
It's all about being able to go home the next morning... right?
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:53am
-
I had the chance to listen to Pat Johnson a couple of times share what he experienced. Very powerful message. One thing that always stuck with me was his description of why we train. He said that the purpose of training is to develop that 6th sense that you feel in your gut, that difference between doing something right or wrong. That's what it's all about. He shared that he didn't listen to the alarms going off in his head. We all need to heed his advice and listen to that voice inside you that acts as a warning if you know how to listen.
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:56am
-
please share what I post with others so we can all learn more about our profession. ms
-
Permalink Reply by Mike Schlags (Captain Busy) Retd on September 27, 2008 at 4:57am
-
the smell alone is gagging, it's the "amines" that are given off... or in other words, the smell of death. bad stuff TJ.. TCSS, Mike
Specialty Websites
Find Members Fast
Firefighting Videos
© 2026 Created by Firefighter Nation WebChief.
Powered by
![]()
Badges | Contact Firefighter Nation | Privacy Policy | Terms of Service